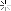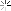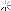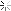Hydrogen Booster
ORIGINAL: omenology
The idea is that you are running the device with the extra power created by the alternator. But, I don't think it would offer any benefits whatsoever to put on a larger alternator, although that would certainly be an idea if one were so inclined. I think I might just try to run it anyway, but who knows how well it'll work out. Oh well.
The idea is that you are running the device with the extra power created by the alternator. But, I don't think it would offer any benefits whatsoever to put on a larger alternator, although that would certainly be an idea if one were so inclined. I think I might just try to run it anyway, but who knows how well it'll work out. Oh well.
Nothing is free, no, but there is definitely waste electricity that is created by any alternator system. Once the battery is charged all that excess power created is simply dissipated as heat because there's no work that can be done with it. The system definitely works, but it needs a much more powerful alternator for it to be efficient in any regard.
Beyond that, it's also fun to do mods that no one or almost no one else has done. I watched a video of the Daytona Bike Rally and it looked like a bike on there actually had a Hydrogen Booster on it. It had 2 seperate tanks and it had an LCD screen with what looked like "H2O" readings on it. So I don't know if it would actually be a one-of-a-kind project, but unique either way.
The reason for doing this is simple, cheap fuel.I have looked into this a little in the past for cheap power and I have found a lot of information. As far as the exact specs I dont have anything that would be useful for you. However you may want to check into 'Joe cells' there is a lot of information out there on how to build your own H2 generators. You can put one together for less than $50 which is a far site better than the 900 that the site posted had. You will need to figure out voltages, H2 output and the water chemistry. If you are only using it on highway miles you shouldnt have to worry about storing the H2 you should be able to just feed it into the fuel/air system directly. I do know thats there are kits available to convert conventional cars over to hydrogen. So unless the BTU's go way higher with the addition of H2 to the gas-air mixed you shouldnt have any problems, though you may have to re-jet your carbs or at least adjust them.
Sorry had to ressurrect this thread cause the science of this thing and the components are just stupidly bad...
OK... the claimed production level on front of webpage 1.5-2l of Browns' gas (2H2O2)per MINUTE
... To produce Browns' gas it requires 193038 Coulombs of power at 1.7VDC per mole of pure water at a pressure of 25psi (otherwise it just reacts w/ itself back to H20 and H2O2 hydrogen peroxide)...then we have some math and a base rate at 1.7VDC@5 amps you will produce 1.678g of gas per Hour at 25psi ....
to get the claimed rate you would need 90 times this base rate, which you can most easily do by upping the Amperage... = 450A of power
So to do this you'd need to maintain the 25psi pressure in the cylinder while drawing off gas (so some kind of pump/compressor system), plus need 450A of current at 1.7v (765W continuous) to produce the required gas. A standard car alternator produces about 150A of useable electricity (on top of cars internal demand)... only real expensive high output diesel models run to 250A... ok... we seeing something off yet?
And this is all going to be done by whats in the picture!?!?!?!?!?!? MY DOG! They must be electro-chemical/physics geniuses to have figured out how to make wattage out of nothing AND to convert water to pressurized gas in a capped PVC tube at 25Psi and run it through 4mm clear flex tubing (its fishtank air/water hosing in the pic not presurized gas line and is fine gas permiable btw... looks like they throw in a bottle of FI cleaner and octane boost fuel additives and the metal thing in the middle is a copper inline thermometer )
)
Sorry to burst your bubble Omenology, but if you took highschool chemistry you'd see this is extremely flawed to say the least.
*edited for gramaticals
OK... the claimed production level on front of webpage 1.5-2l of Browns' gas (2H2O2)per MINUTE
... To produce Browns' gas it requires 193038 Coulombs of power at 1.7VDC per mole of pure water at a pressure of 25psi (otherwise it just reacts w/ itself back to H20 and H2O2 hydrogen peroxide)...then we have some math and a base rate at 1.7VDC@5 amps you will produce 1.678g of gas per Hour at 25psi ....
to get the claimed rate you would need 90 times this base rate, which you can most easily do by upping the Amperage... = 450A of power
So to do this you'd need to maintain the 25psi pressure in the cylinder while drawing off gas (so some kind of pump/compressor system), plus need 450A of current at 1.7v (765W continuous) to produce the required gas. A standard car alternator produces about 150A of useable electricity (on top of cars internal demand)... only real expensive high output diesel models run to 250A... ok... we seeing something off yet?
And this is all going to be done by whats in the picture!?!?!?!?!?!? MY DOG! They must be electro-chemical/physics geniuses to have figured out how to make wattage out of nothing AND to convert water to pressurized gas in a capped PVC tube at 25Psi and run it through 4mm clear flex tubing (its fishtank air/water hosing in the pic not presurized gas line and is fine gas permiable btw... looks like they throw in a bottle of FI cleaner and octane boost fuel additives and the metal thing in the middle is a copper inline thermometer
 )
) Sorry to burst your bubble Omenology, but if you took highschool chemistry you'd see this is extremely flawed to say the least.
*edited for gramaticals
Im gonna t ake you using big words infront of me as a insult...haha just kidding.... looks like either this man threw a bunch of complex things infront of us to confuse us or he knows what hes talking about..So ima leave it at BUSTED!!! But a good idea none the less.
I've actually taken three semesters of college chemistry thus far. And, although I don't have anything in front of me right now, it seems to me that although the H2 + O2 reaction is exothermic, the big thing to consider here is the rate of production and the rate of consumption. The H2 and O2 are not going to recombine into H2O instantly and at the rate that air is being pulled through and into the cylinders I don't think there's ample time for more than a small amount of the gases to recombine. Eh, I could run the reaction rate equations, but I'm kind of tired tonight. Maybe tomorrow.
Oh, and no I wasn't going by any of the given numbers on the webpage nor was I looking to buy what they were selling. It is quite easy to build an apparatus like they have on the cheap.
Oh, and no I wasn't going by any of the given numbers on the webpage nor was I looking to buy what they were selling. It is quite easy to build an apparatus like they have on the cheap.
Thread
Thread Starter
Forum
Replies
Last Post